
A Highly Stable Short α-Helix Constrained by a Main-Chain Hydrogen-Bond Surrogate | Journal of the American Chemical Society
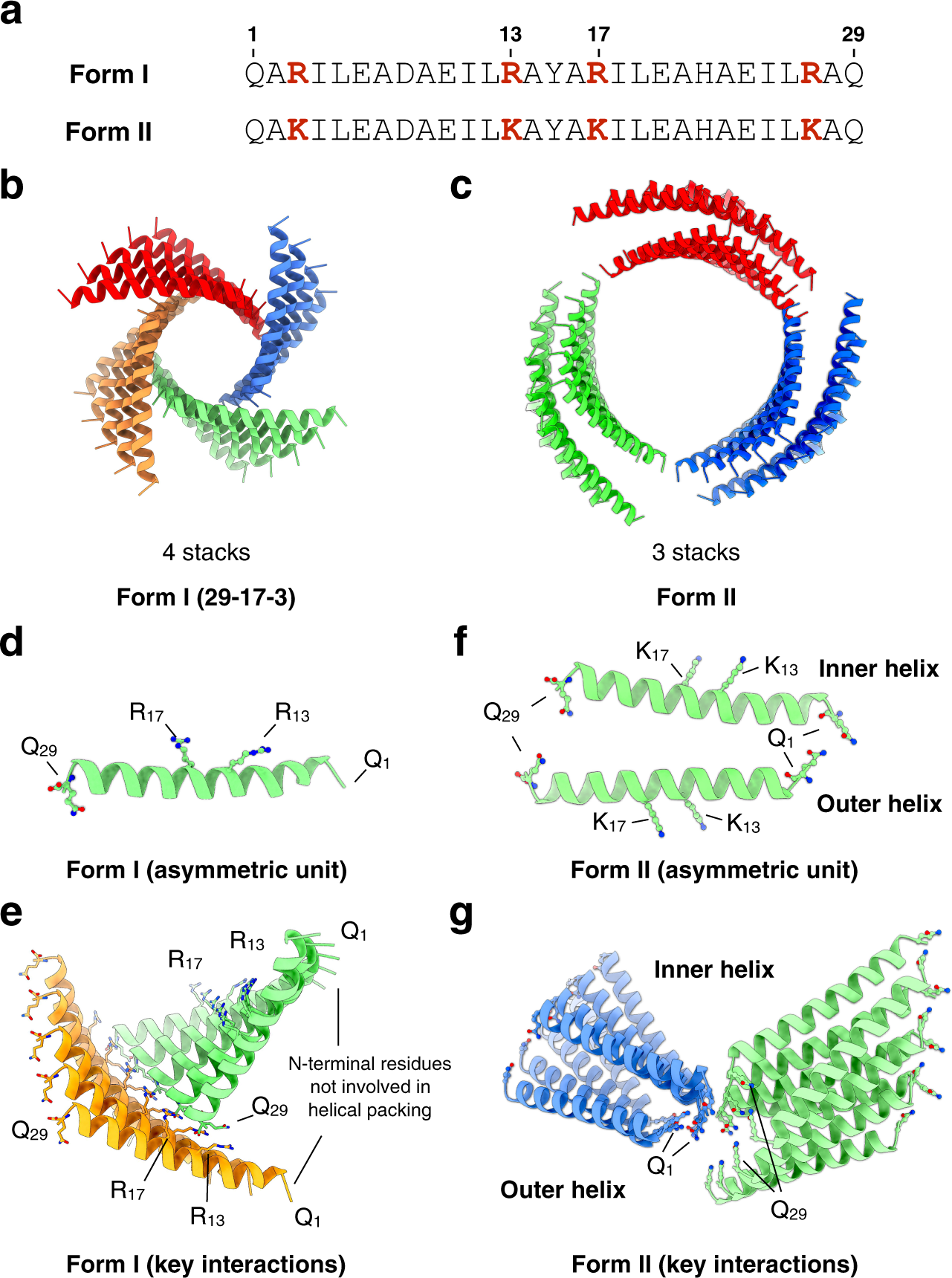
Structural analysis of cross α-helical nanotubes provides insight into the designability of filamentous peptide nanomaterials | Nature Communications
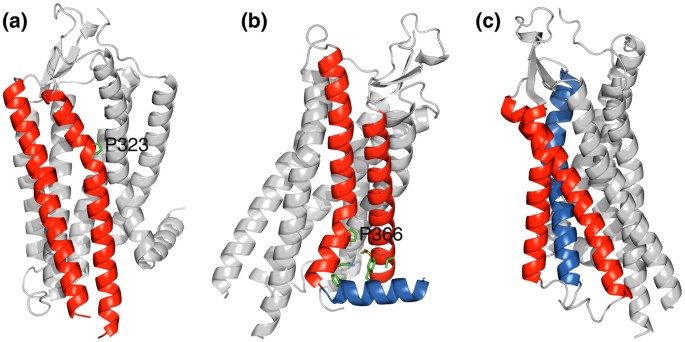
Structural and thermodynamic basis of proline-induced transmembrane complex stabilization | Scientific Reports







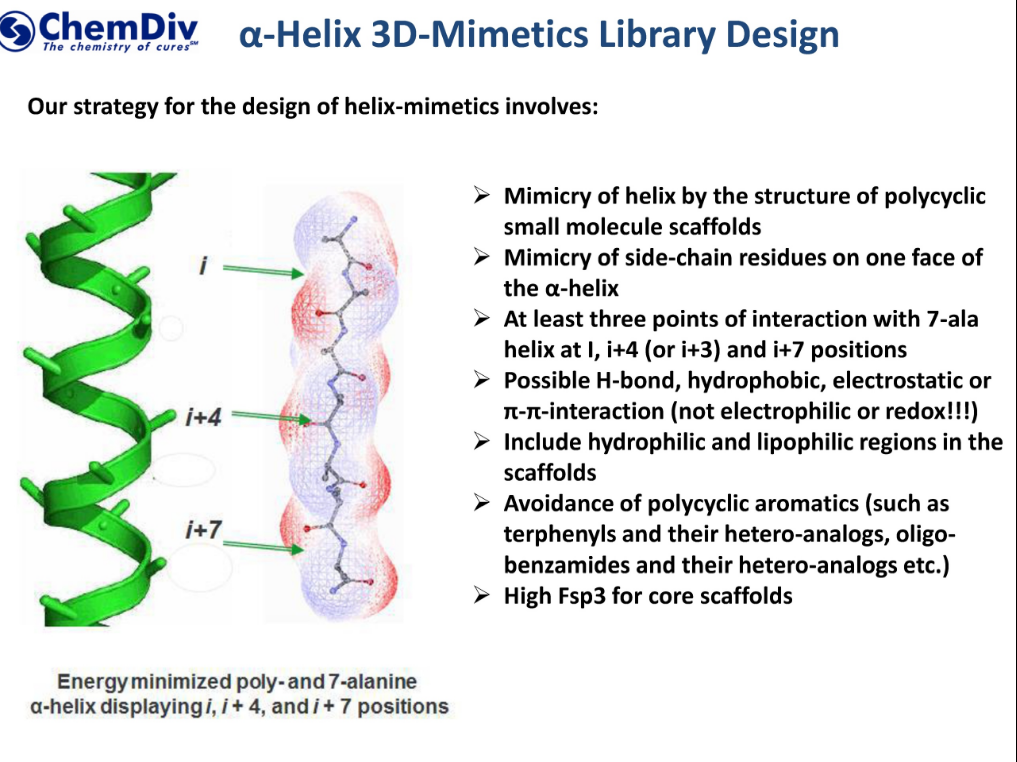



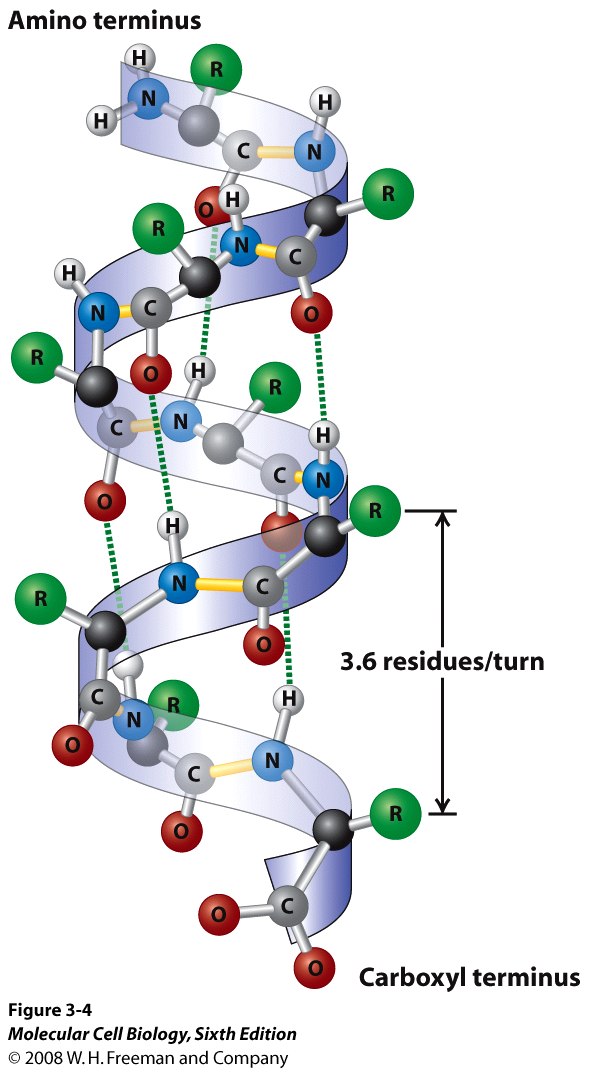
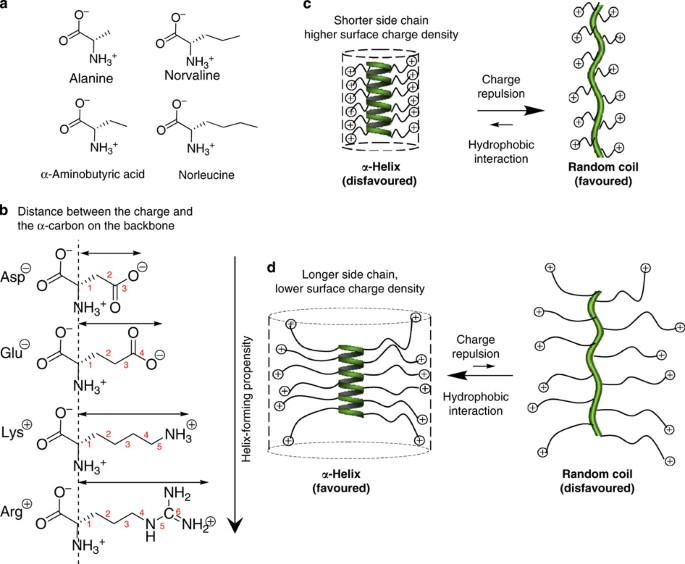
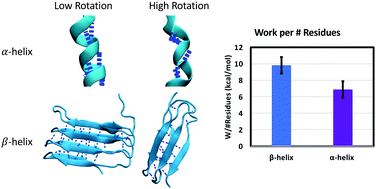

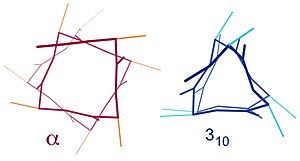




![α-helix structure [6]. | Download Scientific Diagram α-helix structure [6]. | Download Scientific Diagram](https://www.researchgate.net/publication/324706585/figure/fig26/AS:618708808499220@1524523083724/a-helix-structure-6.png)
